Volume 6, Issue 3 (Journal of Clinical and Basic Research (JCBR) 2022)
jcbr 2022, 6(3): 17-22 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Ebrahimi M, Esmaeili H. Frequency and Diversity of Congenital Heart Disease in Children in Northern Iran. jcbr 2022; 6 (3) :17-22
URL: http://jcbr.goums.ac.ir/article-1-363-en.html
URL: http://jcbr.goums.ac.ir/article-1-363-en.html
1- Department of Pediatrics, School of Medicine, Neonatal and Children's Health Research Center, Golestan University of Medical Sciences, Gorgan, Iran , m1355ebrahimi@gmail.com
2- Department of Pediatrics, School of Medicine, Gorgan congenital malformations Research Center, Golestan University of Medical Sciences, Gorgan, Iran.
2- Department of Pediatrics, School of Medicine, Gorgan congenital malformations Research Center, Golestan University of Medical Sciences, Gorgan, Iran.
Keywords: Cardiac anomalies, ventricular septal defect, atrial septal defect, Congenital Heart Diseases
Full-Text [PDF 493 kb]
(1069 Downloads)
| Abstract (HTML) (2240 Views)
Full-Text: (713 Views)
Introduction
Congenital malformations are a major cause of child mortality (1,2). Congenital heart disease (CHD) is the most prevalent congenital anomaly globally, affecting millions of babies per year (3). Previous research on CHD documented congenital abnormalities of 6 to 8 per 1000 live births (4,5), while recent studies reported around 1% of live births. As a result, CHD is the most common congenital disability worldwide (6,7).
Mitchell et al. described CHD as a macroscopic structural abnormality of the heart or large vessels within the chest with severe or possible functional implications (8). While genetic syndromes, teratogenic toxicity, or maternal diabetes may be accounted for around 20% of CHD, there is uncertainty about risk factors for the remaining 80% (9). Various parts of the fetus's heart and other organs are formed from the second to the tenth week of pregnancy. The above factors can cause anatomical disorders in the heart and other organs of the body during this time, disrupting fetal heart function (10).
Valvular heart disease, interventricular septal defect, foramen ovale retention, ductus arteriosus retention, myocardial infarction abnormality, aortic coarctation, Epstein's anomaly, pulmonary artery stenosis, and Tetralogy of Fallot (ToF) are the most common forms of CHD (3,11,12). Since CHD frequently worsens rapidly in infants and children, early detection is critical for a better prognosis following therapy or surgery (8). This study aimed to examine the demographic and clinical characteristics of children with congenital heart anomalies referred to Taleghani Hospital in Gorgan (Iran) between 2014 and 2016.
Materials And Methods
Study sample size and data gathering
From 2014 to 2016, all children referred to Taleghani Hospital in Gorgan were studied in this cross-sectional descriptive study. Census sampling was used. All children were examined by a pediatrician, and those suspected of having CHD were referred to a cardiologist for echocardiography. Children who had an echocardiogram and were found to have congenital heart defects were studied.
Demographic and medical information including gender, gestational age, parent-child relationship, family history of congenital diseases, ethnicity, and any congenital heart defect were retrieved from the patients' records. A VIVID3 echocardiograph (GE, USA) with 3s and 7s probes was used as a measuring device. Furthermore, all children with acquired (non-congenital) heart disease were excluded from the study.
Ethical considerations
Written informed consent was obtained from legally authorized representatives/parents/guardians. All private information of the patients remained confidential during the study. The study received approval from the Ethics Committee of Golestan University of Medical Sciences (approval code: IR.GOUMS.REC.1395.263).
Statistical analysis
The collected data were entered into the SPSS16 software. The Chi-square test was used for comparing the prevalence of CHD types among patients. A p-value less than 0.05 was considered significant
Results
A total of 1,416 patients (572 males and 844 females) were enrolled in the study. At the time of the first echocardiogram, the average age of the subjects was 28.54±13.43 months. Of 1,416 patients, 1272 (89.83%) were at gestational age, while 384 (27.12%) were preterm. Among children at the gestational age, 408 cases (32.1%) had a positive family history of CHD and 864 cases (67.9%) had no family relationship.
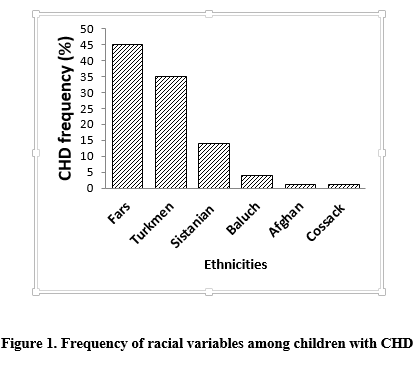
The majority of cases (44.7%) were Fars (Figure 1). Transposition of the great arteries (TGA) was significantly more prevalent in male children than in female counterparts (p=0.028) (Table 1). There was no significant difference in the prevalence of other CHDs between male and female sub
Congenital malformations are a major cause of child mortality (1,2). Congenital heart disease (CHD) is the most prevalent congenital anomaly globally, affecting millions of babies per year (3). Previous research on CHD documented congenital abnormalities of 6 to 8 per 1000 live births (4,5), while recent studies reported around 1% of live births. As a result, CHD is the most common congenital disability worldwide (6,7).
Mitchell et al. described CHD as a macroscopic structural abnormality of the heart or large vessels within the chest with severe or possible functional implications (8). While genetic syndromes, teratogenic toxicity, or maternal diabetes may be accounted for around 20% of CHD, there is uncertainty about risk factors for the remaining 80% (9). Various parts of the fetus's heart and other organs are formed from the second to the tenth week of pregnancy. The above factors can cause anatomical disorders in the heart and other organs of the body during this time, disrupting fetal heart function (10).
Valvular heart disease, interventricular septal defect, foramen ovale retention, ductus arteriosus retention, myocardial infarction abnormality, aortic coarctation, Epstein's anomaly, pulmonary artery stenosis, and Tetralogy of Fallot (ToF) are the most common forms of CHD (3,11,12). Since CHD frequently worsens rapidly in infants and children, early detection is critical for a better prognosis following therapy or surgery (8). This study aimed to examine the demographic and clinical characteristics of children with congenital heart anomalies referred to Taleghani Hospital in Gorgan (Iran) between 2014 and 2016.
Materials And Methods
Study sample size and data gathering
From 2014 to 2016, all children referred to Taleghani Hospital in Gorgan were studied in this cross-sectional descriptive study. Census sampling was used. All children were examined by a pediatrician, and those suspected of having CHD were referred to a cardiologist for echocardiography. Children who had an echocardiogram and were found to have congenital heart defects were studied.
Demographic and medical information including gender, gestational age, parent-child relationship, family history of congenital diseases, ethnicity, and any congenital heart defect were retrieved from the patients' records. A VIVID3 echocardiograph (GE, USA) with 3s and 7s probes was used as a measuring device. Furthermore, all children with acquired (non-congenital) heart disease were excluded from the study.
Ethical considerations
Written informed consent was obtained from legally authorized representatives/parents/guardians. All private information of the patients remained confidential during the study. The study received approval from the Ethics Committee of Golestan University of Medical Sciences (approval code: IR.GOUMS.REC.1395.263).
Statistical analysis
The collected data were entered into the SPSS16 software. The Chi-square test was used for comparing the prevalence of CHD types among patients. A p-value less than 0.05 was considered significant
Results
A total of 1,416 patients (572 males and 844 females) were enrolled in the study. At the time of the first echocardiogram, the average age of the subjects was 28.54±13.43 months. Of 1,416 patients, 1272 (89.83%) were at gestational age, while 384 (27.12%) were preterm. Among children at the gestational age, 408 cases (32.1%) had a positive family history of CHD and 864 cases (67.9%) had no family relationship.
The majority of cases (44.7%) were Fars (Figure 1). Transposition of the great arteries (TGA) was significantly more prevalent in male children than in female counterparts (p=0.028) (Table 1). There was no significant difference in the prevalence of other CHDs between male and female sub

Discussion
The study aimed to assess the prevalence of CHDs in children referred to the Taleghani Hospital in Gorgan (Iran) and to update previous studies by Nikyar et al. (13,14). According to the present study, VSD, ASD, and PDA were the most common types of CHD. In a previous study by Nikyar et al. (2011), ASD was identified as the most common form of heart disorder (13). Other studies in Iran reported similar
results. However, in the present study, newer diagnostic tools were utilized to report the prevalence of CHD types among children. Hematyar et al. reported VSD, ASD, and PDA as the most common CHDs in Tehran (15). However, studies in other parts of Iran reported ASD, ToF, and PDA as the most prevalent types of CHD (16,18). Globally, VSD is the most common CHD, with a prevalence of 22% in Japan (19), 7.62% in China (20), 6.46% in Nigeria (21), 8.41% in the United States (22), 7.45% in Iceland (23), and 9.33% in Saudi Arabia (24). The research population, method of classification, and selection of different criteria, such as live birth and stillbirth, age of diagnosis, diagnostic procedures, and racial and ethnic differences may all contribute to the difference in the prevalence of CHDs around the world.
In this report, female children made up 55% of the patients. The prevalence of TGA was higher in male patients, but the prevalence of other CHDs did not differ significantly between the sexes. Previous studies have also demonstrated that male children are more likely to develop TGA (14, 17, 25). In Saudi Arabia (24) and Iceland (23), however, the prevalence of TGA disorder was similar in both sexes. Since the study's target population was children between the age of 1 and 2 years, cases that died at a younger age could have been excluded, making the study's findings inconsistent with previous research.
Preterm birth was found in 17.7% of children with CHD, which is consistent with the findings of a previous study (26, 27). The difference between the findings of the present study and previous studies may be related to the increased mortality rate among preterm infants born with cardiovascular abnormalities and the severity of their defects, which resulted in death before accessing medical facilities and undergoing echocardiography.
Several critically ill children died during the early stages of resuscitation and before echocardiography, and some minor abnormalities were not detectable in infancy, which could be a limitation of this study. Furthermore, we could not conduct a more in-depth analysis of the prevalence of CHD risk factors among patients due to the paucity of clinical data from patients of different ethnicities. More extensive studies on larger populations are recommended to clarify the impact of other variables, such as geographical area, culture, and socioeconomic status on CHD incidence.
Conclusion
The present study expands our knowledge of the regional frequency of CHD types in children with no acquired heart diseases in Gorgan, Iran. Moreover, VSD is the most common type of CHD among patients, mainly males, followed by ASD and PDA. The highest rate of CHD was observed among Fars patients, which was due to the unmatched numbers in different ethnic groups.
Acknowledgements
The authors would like to thank the Vice-Chancellor for Research and Technology of Golestan University of Medical Sciences and the staff of Taleghani Hospital, the patients, and all the loved ones who helped us in this research.
Declarations:
Funding
Not applicable.
Ethics approvals and consent to participate
The Ethical committee also approves the study of Golestan University of Medical Sciences, Gorgan, Iran, with the approval code of IR.GOUMS.REC.1395.263.
Conflict of interest
The authors declare that they have no competing interests.
Authors' contributions
ME gathered, analyzed, and interpreted the patient data regarding the CHD in children. ME and HE both prepared and revised the final manuscript.
The study aimed to assess the prevalence of CHDs in children referred to the Taleghani Hospital in Gorgan (Iran) and to update previous studies by Nikyar et al. (13,14). According to the present study, VSD, ASD, and PDA were the most common types of CHD. In a previous study by Nikyar et al. (2011), ASD was identified as the most common form of heart disorder (13). Other studies in Iran reported similar
results. However, in the present study, newer diagnostic tools were utilized to report the prevalence of CHD types among children. Hematyar et al. reported VSD, ASD, and PDA as the most common CHDs in Tehran (15). However, studies in other parts of Iran reported ASD, ToF, and PDA as the most prevalent types of CHD (16,18). Globally, VSD is the most common CHD, with a prevalence of 22% in Japan (19), 7.62% in China (20), 6.46% in Nigeria (21), 8.41% in the United States (22), 7.45% in Iceland (23), and 9.33% in Saudi Arabia (24). The research population, method of classification, and selection of different criteria, such as live birth and stillbirth, age of diagnosis, diagnostic procedures, and racial and ethnic differences may all contribute to the difference in the prevalence of CHDs around the world.
In this report, female children made up 55% of the patients. The prevalence of TGA was higher in male patients, but the prevalence of other CHDs did not differ significantly between the sexes. Previous studies have also demonstrated that male children are more likely to develop TGA (14, 17, 25). In Saudi Arabia (24) and Iceland (23), however, the prevalence of TGA disorder was similar in both sexes. Since the study's target population was children between the age of 1 and 2 years, cases that died at a younger age could have been excluded, making the study's findings inconsistent with previous research.
Preterm birth was found in 17.7% of children with CHD, which is consistent with the findings of a previous study (26, 27). The difference between the findings of the present study and previous studies may be related to the increased mortality rate among preterm infants born with cardiovascular abnormalities and the severity of their defects, which resulted in death before accessing medical facilities and undergoing echocardiography.
Several critically ill children died during the early stages of resuscitation and before echocardiography, and some minor abnormalities were not detectable in infancy, which could be a limitation of this study. Furthermore, we could not conduct a more in-depth analysis of the prevalence of CHD risk factors among patients due to the paucity of clinical data from patients of different ethnicities. More extensive studies on larger populations are recommended to clarify the impact of other variables, such as geographical area, culture, and socioeconomic status on CHD incidence.
Conclusion
The present study expands our knowledge of the regional frequency of CHD types in children with no acquired heart diseases in Gorgan, Iran. Moreover, VSD is the most common type of CHD among patients, mainly males, followed by ASD and PDA. The highest rate of CHD was observed among Fars patients, which was due to the unmatched numbers in different ethnic groups.
Acknowledgements
The authors would like to thank the Vice-Chancellor for Research and Technology of Golestan University of Medical Sciences and the staff of Taleghani Hospital, the patients, and all the loved ones who helped us in this research.
Declarations:
Funding
Not applicable.
Ethics approvals and consent to participate
The Ethical committee also approves the study of Golestan University of Medical Sciences, Gorgan, Iran, with the approval code of IR.GOUMS.REC.1395.263.
Conflict of interest
The authors declare that they have no competing interests.
Authors' contributions
ME gathered, analyzed, and interpreted the patient data regarding the CHD in children. ME and HE both prepared and revised the final manuscript.
Article Type: Research |
Subject:
Pediatrics
References
1. Cui H, He C, Kang L, Li Q, Miao L, Shen L, Zhu J, Li X, Wang Y. Under-5-years child mortality due to congenital anomalies: A retrospective study in urban and rural China in 1996− 2013. American Journal of Preventive Medicine. 2016 May 1;50(5):663-71. [View at Publisher] [DOI] [PMID] [Google Scholar]
2. Lee K sun, Khoshnood B, Chen L, Wall SN, Cromie WJ, Mittendorf RL. Infant mortality from congenital malformations in the United States, 1970-1997. Obstet Gynecol. 2001;98(4):620-7.
https://doi.org/10.1016/S0029-7844(01)01507-1 [View at Publisher] [DOI] [PMID] [Google Scholar]
3. Van Der Linde D, Konings EEM, Slager MA, Witsenburg M, Helbing WA, Takkenberg JJM, et al. Birth prevalence of congenital heart disease worldwide: A systematic review and meta-analysis. J Am Coll Cardiol. 2011;58(21):2241-7. [View at Publisher] [DOI] [PMID] [Google Scholar]
4. Dolk H, Loane M, Garne E. Congenital heart defects in Europe: Prevalence and perinatal mortality, 2000 to 2005. Circulation. 2011;123(8):841-9. [View at Publisher] [DOI] [PMID] [Google Scholar]
5. Mir AB, Ahmed K, Jan M, Radakrishnan S. Spectrum of congenital heart disease in a tertiary care centre of Northern India. Int J Contemp Pediatr. 2019;6(3):927. [View at Publisher] [DOI] [Google Scholar]
6. Krasuski RA, Bashore TM. Congenital Heart Disease Epidemiology in the United States: Blindly Feeling for the Charging Elephant. Circulation. 2016;134(2):110-3. [View at Publisher] [DOI] [PMID] [Google Scholar]
7. Meberg A. Congenital heart defects through 30 years. Open J Pediatr. 2012;02(03):219-27. [View at Publisher] [DOI] [Google Scholar]
8. Hoffman JIE, Kaplan S. The incidence of congenital heart disease. J Am Coll Cardiol. 2002;39(12):1890-900. [View at Publisher] [DOI] [PMID] [Google Scholar]
9. Blue GM, Kirk EP, Sholler GF, Harvey RP, Winlaw DS. Congenital heart disease: Current knowledge about causes and inheritance. Med J Aust. 2012;197(3):155-9. [View at Publisher] [DOI] [PMID] [Google Scholar]
10. Marino R V. Nelson Essentials of Pediatrics. Vol. 99, Journal of Osteopathic Medicine. Elsevier; 1999. 23a-23 p. [DOI] [Google Scholar]
11. Brida M, Gatzoulis MA. Adult congenital heart disease: Past, present and future. Acta Paediatr Int J Paediatr. 2019;108(10):1757-64. [View at Publisher] [DOI] [PMID] [Google Scholar]
12. Ejim E, Ike S, Nwaneli U, Oguanobi N, Ubani-Ukoma B, Anisiuba B, et al. Congenital heart diseases in adults: A review of echocardiogram records in Enugu, South-East Nigeria. Ann Med Health Sci Res. 2014;4(4):522. [View at Publisher] [DOI] [PMID] [PMCID] [Google Scholar]
13. Nikyar B, Sedehi M, Mirfazeli A, Qorbani M, Golalipour MJ. Prevalence and pattern of congenital heart disease among neonates in Gorgan, Northern Iran (2007-2008). Iranian journal of pediatrics. 2011 Sep;21(3):307. [Google Scholar]
14. Nikyar B, Sedehi M, Qorbani M, Nikyar A, Golalipour MJ. Ethnical variations in the incidence of congenital heart defects in Gorgan, Northern Iran: A single-center study. The Journal of Tehran University Heart Center. 2014 Jan 1;9(1):9. [Google Scholar]
15. Hematyar M, Khajouie P. Prevalence of congenital anomalies in 1000 live births in Javaheri Hospital, Tehran, 2004. Medical Science Journal of Islamic Azad Univesity-Tehran Medical Branch. 2005 Jun 10;15(2):75-8. [View at Publisher] [Google Scholar]
16. MovahedianA NA, Mosabebi Z, Mazouchi T, Mousavi Q. Prevalence of congenital heart disease in infants admitted to the hospital shahid Beheshti Kashan. Feyz. 2001;18:76-80. [Google Scholar]
17. Mohsenzadeh A, Saket S, Ahmadipour S, Baharvand B. Prevalence and types of congenital heart disease in newborns Khorramabad in2006-2011. Lorestan. 2011;15(5):23-30. [Google Scholar]
18. Song MS, Hu A, Dyhamenahali U, Chitayat D, Winsor EJT, Ryan G, et al. Erratum (Retracted article): Extracardiac lesions and chromosomal abnormalities associated with major fetal heart defects: Comparison. Ultrasound Obstet Gynecol. 2010;35(3):376. [DOI]
19. Takami T. Prevalence of cardiac murmur detected on routine neonatal examination. J Tokyo Med Univ. 2001;59:290-3. [View at Publisher] [Google Scholar]
20. Zhang Y, Riehle-Colarusso T, Correa A, Li S, Feng X, Gindler J, et al. Observed prevalence of congenital heart defects from a surveillance study in China. J Ultrasound Med. 2011;30(7):989-95. [View at Publisher] [DOI] [PMID] [PMCID] [Google Scholar]
21. Sadoh WE, Uzodimma CC, Daniels Q. Congenital Heart Disease in Nigerian Children: A Multicenter Echocardiographic Study. World J Pediatr Congenit Hear Surg. 2013;4(2):172-6. [View at Publisher] [DOI] [PMID] [Google Scholar]
22. Relier MD, Strickland MJ, Riehle-Colarusso T, Mahle WT, Correa A. Prevalence of congenital heart defects in metropolitan Atlanta, 1998-2005. Obstet Gynecol Surv. 2009;64(3):156-7. [DOI]
23. Stephensen SS, Sigfusson G, Eiriksson H, Sverrisson JT, Torfason B, Haraldsson A, et al. Congenital cardiac malformations in Iceland from 1990 through 1999. Cardiol Young. 2004;14(4):396-401. [View at Publisher] [DOI] [PMID] [Google Scholar]
24. Alabdulgader AAA. Congenital heart disease in Saudi Arabia: Current epidemiology and future projections. East Mediterr Heal J. 2006;12(SUPPL. 2). [View at Publisher] [Google Scholar]
25. Animasahun BA, Madise-Wobo AD, Gbelee HO, Omokhodion SI. Children with transposition of the great arteries: Should they actually be born in Nigeria?. World Journal of Cardiology. 2017 Mar 26;9(3):277. [DOI] [PMID] [PMCID] [Google Scholar]
26. MUIR CS. Incidence of congenital heart disease in Singapore. Br Heart J. 1960;22(1):243-54. [DOI] [PMID] [PMCID] [Google Scholar]
27. Tanner K, Sabrine N, Wren C. Cardiovascular malformations among preterm infants. Pediatrics. 2005;116(6):e833-8. [View at Publisher] [DOI] [PMID] [Google Scholar]
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International (CC BY-NC 4.0).





